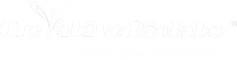The landscape of therapeutic antibodies is rapidly evolving. For decades, the biopharmaceutical industry has heavily relied on the standard Immunoglobulin G (IgG) framework to develop life-saving treatments for oncology, autoimmune diseases, and infectious diseases. However, as the limitations of IgG become more apparent—particularly in mucosal immunity and multivalent avidity—researchers are increasingly turning their attention to non-IgG isotypes, including IgA, IgM, and IgE.
While these non-IgG classes offer remarkable therapeutic potential, they bring unprecedented structural complexities. Chief among these challenges is glycosylation. As a critical post-translational modification (PTM), glycosylation influences everything from structural stability and half-life to effector function and immunogenicity. In non-IgG antibodies, glycosylation is vastly more complex than in standard IgGs, meaning that conventional manufacturing and analytical approaches are no longer sufficient.
In this comprehensive guide, we will explore why glycosylation matters significantly more in non-IgG therapies, identify the primary risk points in development, and outline a practical QC strategy to ensure the safety and efficacy of your next-generation therapeutics.
Why Glycosylation is Uniquely Critical in Non-IgG Antibodies
To understand the heightened importance of glycosylation in non-IgG antibodies, we must look at the fundamental structural differences between these molecules and standard IgGs.
A typical human IgG1 contains a single, highly conserved N-linked glycosylation site in its Fc region (Asn297). This single glycan accounts for only about 2-3% of the total molecular weight of the antibody. In stark contrast, non-IgG antibodies are heavily glycosylated, with carbohydrates comprising anywhere from 7% to over 12% of their total mass.
The Glycosylation Profile of IgA
IgA exists in two subclasses: IgA1 and IgA2. Monomeric IgA contains multiple N-linked glycosylation sites, while IgA1 uniquely features a heavily O-glycosylated hinge region. When IgA forms dimers (secretory IgA), it incorporates a Joining (J) chain and a Secretory Component (SC), both of which introduce even more N-linked glycans. These complex glycan structures are crucial for protecting the antibody from proteolytic degradation in harsh mucosal environments.
The Glycosylation Profile of IgM
IgM is the largest antibody isotype, typically assembling into pentamers or hexamers. A single IgM pentamer, along with its J chain, contains a staggering 51 N-linked glycosylation sites. This massive glycan array is not merely decorative; it is absolutely essential for the proper oligomerization, structural integrity, and potent complement-dependent cytotoxicity (CDC) that makes IgM such an attractive candidate for targeting cancer cells.
The Glycosylation Profile of IgE
Though primarily known for its role in allergic responses, IgE is being explored for targeted cancer therapies (AllergoOncology). IgE contains seven N-linked glycosylation sites across its heavy chains. The glycans on IgE are directly involved in its ultra-high-affinity binding to the FcεRI receptor, dictating its potent biological activity.
Key Risk Points in Non-IgG Glycosylation
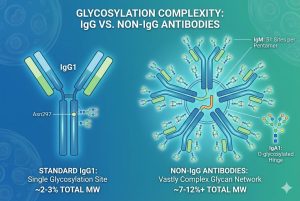
Because the glycan burden in non-IgG antibodies is so high, deviations during cell culture or bioprocessing can lead to severe consequences. Below are the critical risk points that developers must monitor.
1. Extreme Heterogeneity and Batch-to-Batch Inconsistency
With dozens of glycosylation sites on a single multimeric molecule (like IgM), the potential for both macro-heterogeneity (variation in site occupancy) and micro-heterogeneity (variation in the specific glycan structure attached to a site) is astronomical. This massive structural diversity makes it incredibly difficult to achieve batch-to-batch consistency. If manufacturing conditions—such as dissolved oxygen, pH, or nutrient availability—shift even slightly, the resulting glycoform profile can change drastically, leading to a failed batch.
2. Reduced Serum Half-Life and Rapid Clearance
Non-IgG antibodies naturally have shorter serum half-lives than IgGs because they lack strong binding to the neonatal Fc receptor (FcRn). Therefore, their survival in the bloodstream is heavily dependent on their glycosylation profile—specifically, the degree of terminal sialylation. If non-IgG antibodies are produced with exposed, under-sialylated galactose or mannose residues, they will be rapidly recognized and cleared by the Asialoglycoprotein receptor (ASGPR) or mannose receptors in the liver. Failing to optimize sialylation during expression is a major risk point that can render a therapeutic practically useless in vivo.
3. Unintended Immunogenicity
Therapeutic antibodies are typically produced in mammalian cell lines like CHO (Chinese Hamster Ovary) or HEK293. While CHO cells are industry standard, they possess the enzymatic machinery to attach non-human glycan epitopes, such as α-1,3-galactose (alpha-gal) and N-glycolylneuraminic acid (Neu5Gc). Because non-IgG antibodies possess so many glycosylation sites, the statistical probability of accumulating these immunogenic, non-human glycans is much higher than in IgG. Introducing these into a patient can trigger severe anti-drug antibody (ADA) responses or anaphylaxis.
4. Structural Instability and Aggregation
Glycans act as physical spacers and structural stabilizers. In molecules like IgM and IgA, missing glycans can expose hydrophobic patches on the protein backbone, driving protein aggregation. Aggregated proteins not only lose their therapeutic efficacy but are also highly immunogenic.
Establishing a Practical QC Strategy Control
Given the risks outlined above, establishing a robust, multi-tiered QC strategy is non-negotiable for non-IgG therapeutic development. A practical control strategy must encompass both upstream bioprocess controls and advanced downstream analytical characterization.
Upstream Control: Cell Line and Metabolic Engineering
The first line of defense in your QC strategy is preventing poor glycosylation from happening in the first place.
-
Host Cell Selection: Utilize glycoengineered cell lines specifically designed to boost sialylation or eliminate the production of immunogenic alpha-gal and Neu5Gc residues.
-
Media Supplementation: The metabolic pathways that build glycans require specific building blocks. Supplementing bioreactor media with precursors like galactose, uridine, and manganese can push the cellular machinery toward more complete glycan maturation and higher sialic acid content.
Downstream Analytical QC: A Multi-Tiered Approach
Because intact mass analysis is often insufficient for resolving the massive complexity of fully assembled IgM or secretory IgA, your analytical QC strategy must utilize a “divide and conquer” methodology.
Tier 1: Intact and Subunit Analysis Using High-Resolution Mass Spectrometry (HRMS) combined with liquid chromatography (LC), analysts can evaluate the intact monomeric units or enzymatically digested subunits (middle-down approach). This provides a macro-level view of the major glycoforms and ensures that the overall glycosylation profile is within acceptable limits.
Tier 2: Peptide Mapping (Bottom-Up Analysis) To understand exactly what is happening at each specific glycosylation site, bottom-up peptide mapping is required. The antibody is digested into peptides using proteases like trypsin, and analyzed via LC-MS/MS. This allows QC teams to measure site-specific occupancy (macro-heterogeneity) and determine exactly which glycan structures are attached to critical sites, ensuring that residues responsible for receptor binding are correctly modified.
Tier 3: Released Glycan Profiling For precise quantification of the glycan species present, glycans are enzymatically cleaved from the protein backbone (typically using PNGase F for N-glycans), fluorescently labeled (e.g., with 2-AB or procainamide), and analyzed using Hydrophilic Interaction Liquid Chromatography with Fluorescence Detection (HILIC-UHPLC-FLD). This is the gold standard for monitoring the relative abundance of critical glycan features, such as the total percentage of sialylation, fucosylation, and high-mannose structures.
Defining Critical Quality Attributes (CQAs)
A practical QC strategy relies on establishing strict acceptance criteria. For non-IgG antibodies, developers must define specific glycosylation features as Critical Quality Attributes (CQAs). For example, a CQA for a therapeutic IgM might require >80% terminal sialylation to ensure adequate half-life, and <1% high-mannose structures to prevent rapid clearance. These specifications must be tightly monitored throughout the product lifecycle.
Conclusion
As the biopharma industry ventures deeper into the promising realm of non-IgG therapeutics, mastering the complexities of glycosylation is paramount. The dense and varied glycan networks of IgA, IgM, and IgE govern their stability, clearance, and immunological functions. By understanding the unique risk points—from extreme heterogeneity to rapid in vivo clearance—and implementing a rigorous, multi-tiered QC strategy, developers can successfully navigate these challenges and bring safe, highly efficacious next-generation therapeutics to the clinic.
Accelerate Your Non-IgG Therapeutic Programs
At Creative Biolabs, we specialize in the unique challenges of non-IgG antibody development and characterization. If you are looking for expert support to manage complex glycosylation profiles, optimize your cell lines, or establish a robust analytical framework, we are here to help.
Explore our specialized services below: